Internal Rotor Input in .mrt File
Following is the content of sample .mrt file,
 Each line corresponds to a internal rotor, with a format;
Each line corresponds to a internal rotor, with a format;
iprR100.mrt;1:3-5-6-7 1:4-8-9-10

pivot-moi2:moi1The
moi1 input is the same as that in
gpop6irt
program, while the moiety-2 is specified by the pivot atom only.
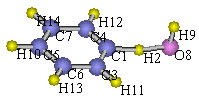
The sample input specifies the rotation around C[1]–C[3]
bond and that around C[1]–C[4] bond.